Erasmus scientist plays crucial role in genetics breakthrough
 A major development in the world of genetics has boosted hopes of better treatment for hereditary illnesses.
A major development in the world of genetics has boosted hopes of better treatment for hereditary illnesses.
By Moira Holden
Researchers from a Dutch university have discovered vital components of genes which could lead to significant progress in the fight to eradicate genetic conditions. Scientists have attempted to crack these codes for the past 170 years, but now the breakthrough has been achieved.
Tobias Knoch, head of biophysical genomics at Erasmus Medical Centre, Rotterdam, led the study into the structure of genomes, alongside German counterpart Malte Wachsmuth, from the European Molecular Biology Laboratory, Heidelberg. The 20-year-long research has now yielded results which fill in the missing parts of the genetic jigsaw.
Advances
‘This is a big step forward,’ says Knoch. ‘With our discovery we can now decode every level on which genetic information is stored, and, if differences occur, to see what this might mean in the possibility of it leading to a disease. We can now finally understand the organisation and function of genomes much more completely in healthy organisms and how genetic diseases develop.’
The study into the organisation of genes in 3D space developed new biomechanical methods and microscopes, but also set up a world-wide computing grid to meet the demands of the research.
Genome
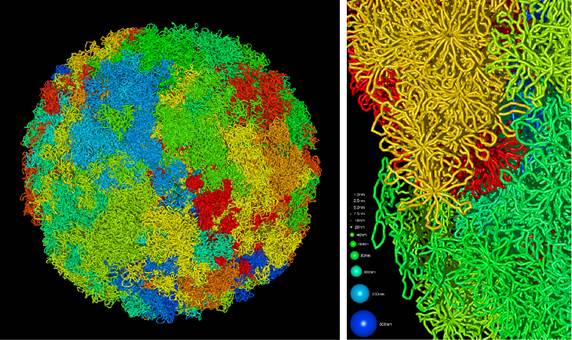
Knoch’s work helped to pin down the missing three blocks out of the eight levels in the eukaryotic genome – scientists had previously revealed five of these eight levels in detail, but the most important three central blocks in the genetic architecture had remained elusive. The new study discovered the role of the chromatin quasi-fibre which folds into small loops to form stable rosettes – this plays a crucial role in genome maintenance and control.
‘Now that we have all structural levels, we can understand genome function completely which was impossible before,’ said Knoch. ‘In principle, we have described the structure of what many people consider to be the missing link in genome research.’
What is a genome? It’s a collection of genes which acts as instructions of the big human machine where all of the information for the building and working of the machine is stored. Knoch compares knowledge of the structure of the genome to the workings of a collection of books with chapters, paragraphs, sentences, words and letters. ‘You would be able to read letters and words, but you are not able to read sentences and paragraphs and would only know that there were different chapters,’ he says.
Diseases
The discovery of the working of the three missing parts now heralds an opportunity to further the medical advance into hereditary conditions. ‘This is not only a very important first step towards final structural genome sequencing, but also for disease diagnostics, treatment, and especially for all genome engineering efforts currently discussed publicly,’ said Knoch.
A genetic disease is any disease caused by an abnormality of a person’s genome in one or more parts genetic make-up. Some genetic conditions are inherited from parents, but other genetic diseases are the result of spontaneous changes and can happen randomly or can be caused by environmental factors.
Most genetic diseases, such as cystic fibrosis, sickle cell anaemia and Huntington’s disease are caused by a single faulty gene. But multifactorial inheritance conditions, such as cancer, Alzheimer’s, arthritis, diabetes, cancer and obesity, are caused by a combination of environmental factors in multiple genes.
Future
Possible treatment for genetic conditions is now a step nearer. The new coding levels mean medical professionals can start looking at all levels to find out where the origin of a disease lies and if treatment can be given.
Prader-Willi syndrome is one disease which could benefit. Babies with the condition have weak muscle tone, feeding difficulties, poor growth and development, and as they grow they tend to over-eat and can have learning disabilities.
Knoch says: ‘Where Prader-Willi is concerned, we have patients where the mutation is on the DNA level, and other patients where the error is on the second level while the first DNA level is okay. We also have cases where we do not know where the error is. Now we can look at the other levels where we might find the missing cause.’
Britain recently passed a law to allow ‘three-parent IVF’ to stop mitochondrial diseases – serious genetic diseases – being passed on to babies. The DNA would come from three people in a pro-nuclear transfer, with the healthy mitochondria from the donor’s egg added to the embryo while the unhealthy mitochondria from the female parent would be removed.
But the process has raised concerns about ethics. ‘The room for agricultural breeding and manipulation might be quite big and has huge opportunities, but currently, as in cases of three-parent babies we should be very careful and be prepared for the fact that the risk might be bigger than we think,’ warns Knoch.
‘We just have described all levels where genetic information is stored and now first have to understand how the system entirely works. It is very likely one solves one issue and right away creates the next, and perhaps even bigger, problem.’
Thank you for donating to DutchNews.nl.
We could not provide the Dutch News service, and keep it free of charge, without the generous support of our readers. Your donations allow us to report on issues you tell us matter, and provide you with a summary of the most important Dutch news each day.
Make a donation